An electrochemical gas sensor reacts with the target gas to produce an electrical current proportional to the gas concentration. The vast majority of devices used for toxic gas detection in industrial facilities rely on electrochemical gas sensors. Their use has become the industry standard for measuring toxic gases such as carbon monoxide (CO), nitrogen dioxide (NO₂), hydrogen sulfide (H₂S), ammonia (NH₃), and ozone (O₃).
However, incorrect sensor selection or a flawed circuit design can directly impact device accuracy, the certification process, and field safety. In this article, we examine step by step the working principle of electrochemical gas sensors, the parameters that guide selection, and the engineering details that directly affect gas detection device development.
1. How Do Electrochemical Gas Sensors Work?
Electrochemical gas sensors produce a current proportional to the target gas concentration as a result of an electrochemical reaction with the target gas. The sensor current output is typically in the nano-to-microampere range (nA – µA).
Working Principle
The working principle of an electrochemical gas sensor can be summarized in four steps:
- Gas diffuses into the sensor
- A reaction occurs at the electrode surface
- Electron transfer takes place
- The sensor produces a current proportional to concentration
Core Components
An electrochemical sensor consists of four fundamental components:
- Diffusion barrier — controls gas entry
- Working electrode — where the target gas reacts
- Reference electrode — maintains a stable potential
- Counter electrode — completes the circuit
Sensor Bias Voltage
The sensor circuit applies a bias voltage between the working and reference electrodes and measures the current between the counter and working electrodes — a current that is proportional to gas concentration. The bias voltage is a controlled potential applied by the potentiostat circuit, and it varies depending on the target gas and sensor type.
| Sensor | Target Gas | Bias Voltage |
|---|---|---|
| SGX-4CO | Carbon Monoxide | 0 mV |
| Winsen ME3-O3 | Oxygen | 0 mV |
| SGX EC410 (O2)* | Oxygen | −600 mV |
| Alphasense NO-A1 | Nitric Oxide | +300 mV |
| SGX-7H2S | Hydrogen Sulfide | 0 mV |
* The −600 mV value is sourced from SGX Sensortech Application Note 2.
Bias voltage directly affects potentiostat circuit design. Sensors with 0 mV bias can operate with a simpler analog front-end, while sensors requiring negative or positive bias demand a stable reference voltage source and a low-offset op-amp selection.
2. Number of Electrodes in Electrochemical Gas Sensors
One of the most critical parameters when selecting an electrochemical sensor is the number of electrodes. Electrode count directly impacts measurement accuracy and cost.
2-Electrode Sensors
These sensors (Figarro TGS5141, ME2-CO-Φ14x5, etc.) share a common counter and reference electrode. At high concentrations, the elevated current causes drift in the sensor bias voltage, affecting measurement accuracy. These sensors (with the exception of 2-electrode oxygen sensors) are generally used in low-cost devices such as residential CO detectors.
3-Electrode Sensors
The majority of toxic gas detection devices on the market use 3-electrode electrochemical sensors. In these sensors (SGX-4NO2, SGX-4CO, Winsen ME3-CO, etc.), the reference and counter electrodes are separate, resulting in higher accuracy and better stability. They are widely used in parking garage gas detection, HVAC, and industrial toxic gas measurement.
Advantages over 2-electrode sensors:
- Better stability
- Lower drift
- Higher accuracy
For this reason, 3-electrode sensors should be the starting point when evaluating sensors for industrial gas detection device development projects.
4-Electrode Sensors (Professional Applications)
There are two distinct 4-electrode electrochemical gas sensor architectures on the market:
- Shared reference and counter electrode sensors
- Auxiliary electrode sensors
Shared Reference and Counter Electrode Sensors
These sensors are a modified version of 3-electrode designs. They feature a shared reference and counter electrode along with two separate working electrodes, enabling a single sensor to detect two different gases simultaneously. Examples include SGX-CO-H2S-LP, Alphasense SOH-A2, and Alphasense COH-A2.
Auxiliary Electrode Sensors
In these sensors (e.g., Alphasense CO-B4, Alphasense SO2-B4), a fourth "Auxiliary" electrode is added alongside the working, counter, and reference electrodes. This fourth electrode has no gas contact; instead, the current measured across it reflects environmental influences. The difference in current between the working and auxiliary electrodes is directly proportional to the target gas concentration.
Advantages:
- High accuracy
- High stability
- Environmental effects minimized via the 4th electrode
These sensors are capable of ppb-level measurement and are primarily used for ambient air quality monitoring.
Sensor Comparison by Electrode Count
| 2-Electrode | 3-Electrode | 4-Electrode | |
|---|---|---|---|
| Accuracy | Low | Medium | High |
| Cost | Low | Medium | High |
| Application | Residential | Industrial | Ambient air quality |
| Example Sensors | Figarro TGS5141, ME2-CO-Φ14x5 | SGX-4CO, SGX-4NO2, Winsen ME3-CO | Alphasense SO2-B4, Alphasense OX-B431 |
3. What Is ppm/nA Sensitivity in Electrochemical Gas Sensors?
The most important parameter to check in a datasheet when selecting an electrochemical gas sensor is the sensitivity value, expressed as nA/ppm. For example, for a sensor with 55 nA/ppm sensitivity under ideal conditions:
| Gas Concentration | Sensor Current |
|---|---|
| 1 ppm | 55 nA |
| 10 ppm | 550 nA |
| 20 ppm | 1.1 µA |
Since output currents are extremely small, leakage currents must be carefully managed during potentiostat circuit design. Ambient temperature also significantly affects sensor sensitivity. The chart below shows the temperature-dependent output variation of the SGX-4CO carbon monoxide sensor.
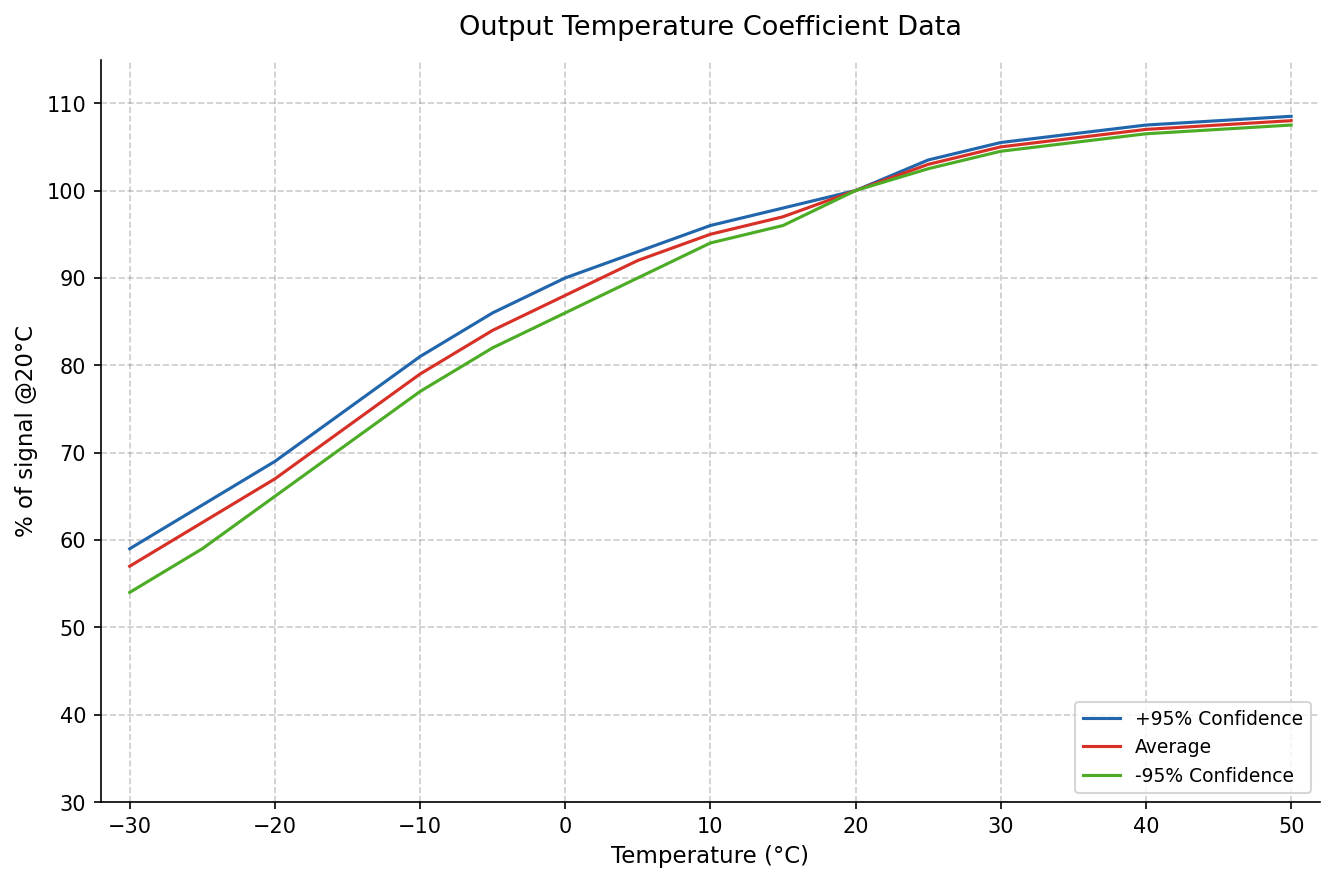
With 20°C as the reference point, sensor output drops to approximately 55% when temperature falls to −30°C. If this deviation is not compensated, the resulting measurement error can translate directly into a safety risk. For this reason, temperature compensation must be an integral part of the circuit design in devices operating across a wide temperature range.
4. T90 Response Time in Electrochemical Gas Sensors
The time required for a gas sensor to reach 90% of the full-scale reading is expressed in the datasheet as T90 (response time). For typical electrochemical sensors, T90 falls within the 20–60s range. However, in real-world applications, T90 depends not only on the sensor itself but also on the enclosure design of the gas detection device.
Factors Affecting T90
- Filters and membranes
- Gas diffusion holes
- Airflow
- Device enclosure design
For example, the same sensor in an open enclosure design may achieve 25s T90, while a filtered, closed enclosure can push that figure to 55–60s. This directly affects pass/fail criteria, particularly in applications where rapid alarm response is required.
For this reason, sensor selection and mechanical design must be addressed together in gas detection device development.
5. Electrochemical Gas Sensor Lifespan and Calibration
Electrochemical gas sensors are consumable components whose performance degrades over time.
Typical values:
- Sensor lifespan: ~2 years
- Calibration interval: 6–12 months
The primary cause of performance degradation in electrochemical sensors is the drying of the electrolyte within the sensor. This process accelerates in high-temperature, low-humidity environments, where span drift increases significantly and more frequent calibration becomes necessary.
Types of Drift Over Time
- Zero drift — shift in the sensor's zero point
- Span drift — change in the measurement curve
- Sensitivity reduction — decrease in output per unit concentration
Using a two-point calibration technique — first a zero calibration with clean air, then a span calibration with a known reference gas concentration — drift effects can be corrected.
For long-term measurement accuracy, the following parameters must be incorporated into the gas detection device design from the outset:
- ✔ Periodic calibration
- ✔ Maintenance plan
- ✔ Sensor replacement strategy
6. Cross Sensitivity and Environmental Effects in Electrochemical Gas Sensors
Electrochemical sensors can respond to gases other than their target gas. For example, an NO₂ sensor may be affected by H₂S.
This can lead to the following risks:
- False alarms
- Certification failure
- Field safety hazards
Cross Sensitivity Table
| Sensor | Target Gas | Interfering Gas | Cross Sensitivity |
|---|---|---|---|
| Alphasense OX-A431 | Ozone | Hydrogen sulfide | −80% |
| Winsen ME3-NO2 | Nitrogen dioxide | Cl₂ | +100% |
| SGX-4SO2 | Sulfur dioxide | Nitrogen dioxide | −100% |
A key observation from the table is the presence of negative cross sensitivity values. A negative cross sensitivity means that the interfering gas causes the sensor's target gas reading to decrease. For example, when H₂S is present, the Alphasense OX-A431's ozone detection drops by 80%. Similarly, in an environment containing both SO₂ and NO₂, if SGX-4SO2 is used for SO₂ measurement, the NO₂ will generate −100% cross sensitivity, causing the SO₂ reading to appear significantly lower than the actual value.
In environments with multiple gases, cross sensitivity values must be accounted for during the design process. Two approaches can be applied:
- Select a sensor with low cross sensitivity — Review datasheets and choose a sensor where interfering gas effects are negligible.
- Use a multi-sensor architecture — Measure each gas in the environment with a dedicated sensor, and compensate for cross effects at the software layer.
Environmental Factors
Additional factors affecting sensor performance:
- Temperature
- Humidity
- Solvents
- Silicone vapor (sensor poisoning)
The application environment must always be analyzed during sensor selection, and appropriate protective measures must be taken against environmental influences.
Field Note: Cross Sensitivity in SPEC Sensors — Findings from TÜBİTAK 1501 Laboratory Tests
During the preliminary research phase of a TÜBİTAK 1501-funded air quality monitoring project, we conducted controlled cross sensitivity experiments using SPEC Sensors electrochemical sensors to evaluate the application of low-cost sensors for air quality monitoring.
CO gas was applied to SPEC Sensors H₂S and SO₂ sensors at varying concentrations. The figure below shows the ADC output of each sensor in response to CO exposure:
Figure 2: Cross sensitivity profile of SPEC Sensors electrochemical sensors to CO gas — TÜBİTAK 1501 Project
Key findings from the experiment:
- H₂S sensor: Produced a measurable response to CO gas. While sensor response to CO gas concentration below 80 ppm was negligible, a remarkable response was observed starting at 80 ppm CO, reaching an ADC value of approximately 3.243M at 200 ppm CO — a deviation of approximately 3.1% relative to the baseline value (≈3.145M).
- SO₂ sensor: Exhibited a significantly higher cross-sensitivity response. The ADC value increased from approximately 1.049M at 80 ppm CO to 1.382M at 200 ppm CO — approximately a 68% increase over the baseline (≈820K). This finding indicates that the effect of CO concentration on SO₂ sensor output is not negligible and must be taken into concentration. If not, it can lead to false measurements in multi-gas environments where both CO and SO₂ are present simultaneously.
These findings were published in our conference paper which examines application of low-cost sensors for air quality monitoring applications.
Practical takeaway: In multi-gas environments, low-cost sensors may not provide sufficient selectivity. In such cases, either select a sensor with a lower cross sensitivity specification, or implement a multi-sensor fusion algorithm at the software level to compensate for interference effects. Based on performance analysis, we moved to Alphasense B4 series sensors.
7. Electrochemical Sensor Selection Checklist
When developing a gas detection device, sensor selection alone is not sufficient. Sensor performance, circuit design, mechanical enclosure design, and calibration strategy must all be addressed as an integrated whole. Key parameters to consider in gas detection device development:
- ✔ Target gas and application area
- ✔ Measurement range
- ✔ Cost
- ✔ Sensitivity value
- ✔ T90 requirement
- ✔ Temperature compensation requirement
- ✔ Sensor lifespan and calibration plan
- ✔ Other gases present in the environment
- ✔ Certification requirements
Correct sensor selection directly affects product accuracy, maintenance costs, and the certification process — reducing long-term device costs. For end-to-end support from sensor selection through circuit design and certification, explore our gas measurement device development service.
For a general comparison of gas sensor technologies, see our article How to Select a Gas Sensor: Gas Sensor Types and Comparison Guide.
In our next article, we will examine how to build an analog front-end architecture for developing gas detection devices with electrochemical gas sensors.
References
- SGX Sensortech. Electrochemical Sensors Application Note 2: Design of Electronics for Electrochemical Gas Sensors. SGX Sensortech Ltd. Link
- SGX Sensortech. SGX-4CO Carbon Monoxide Sensor Datasheet. SGX Sensortech Ltd.
- SGX Sensortech. SGX-7H2S Hydrogen Sulphide Sensor Datasheet. SGX Sensortech Ltd.
- SGX Sensortech. SGX-4SO2 Sulphur Dioxide Sensor Datasheet. SGX Sensortech Ltd.
- Alphasense Ltd. OX-A431 Ozone Sensor Datasheet. Alphasense Ltd.
- Winsen Electronics. ME3-NO2 Nitrogen Dioxide Sensor Datasheet. Zhengzhou Winsen Electronics Technology Co., Ltd.
- Heydarov, S., Faydacı, Ç., Öztürk, S., Kösemen, A., & Aksu, A. (2022). Evaluation of Low Cost Electrochemical and Infrared Sensors in the Aspect of Air Quality Monitoring. ELECO 2022 — 12th International Conference on Electrical and Electronics Engineering, Bursa, Türkiye. Google Scholar
